Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Title
Atezolizumab in patients with urinary tract squamous cell carcinoma: a single arm, open label, multicentre, phase II clinical trial.
Description
AURORA is a phase II open label trial of Atezolizumab in patients with urinary tract squamous cell carcinoma (UTSCC). UTSCC is the most common of the rare urinary tract cancer histologies, comprising 2.1–6.7% of urinary tract cancers overall. There are few data available to guide treatment decisions for UTSCC. The AURORA trial will test the hypothesis that PD-L1 inhibition with atezolizumab immunotherapy is clinically effective, tolerable and safe, in patients with urinary tract squamous cell carcinoma (UTSCC). Translational endpoints will aim to determine characteristics for responsiveness to this treatment. AURORA was developed on behalf of the International Rare Cancers Initiative (IRCI) and the National Cancer Research Institute Bladder and Renal Group.
Outcomes
Primary Outcomes:
- To determine the clinical activity of atezolizumab in patients with incurable histologically confirmed, immunotherapy naïve UTSCC.
- Primary endpoint: Best overall objective response rate (ORR; the percentage with partial (PR) or complete (CR) response) by RECIST v1.1
Secondary Outcomes:
- To determine the safety and tolerability of atezolizumab in this clinical setting.
- To determine the overall survival (OS) of patients treated with atezolizumab in this clinical setting.
- To determine the progression-free survival (PFS) of patients treated with atezolizumab in this clinical setting.
- To determine the duration of response of patients treated with atezolizumab in this clinical setting.
- To determine efficacy by iRECIST as an alternative means of response assessment in this clinical setting.
- To determine tumour volume changes of individual patients treated with atezolizumab in this clinical setting.
- To determine the impact on quality of life of the atezolizumab in this clinical setting.
- To determine the impact of PD-L1 expression status on clinical response.
Trial Design
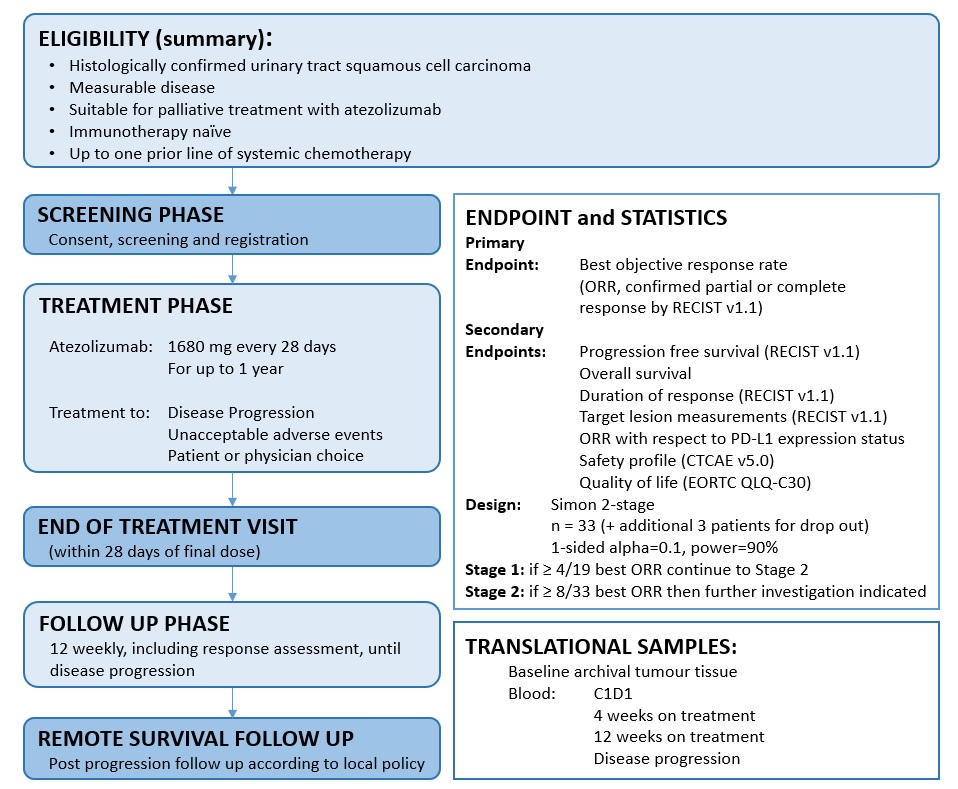
AURORA is a single arm, open label, multicentre, phase II clinical trial of atezolizumab immunotherapy, in immunotherapy naive patients with urinary tract squamous cell carcinoma (UTSCC).
Recruitment is intended to occur over approximately 2 years and will follow a two stage statistical design. However, the intention to allow continuous recruitment between Stage 1 and Stage 2.
Following a Screening Phase of up to 28 days, eligible patients will be registered and will then commence atezolizumab immunotherapy, every 28 days, within a Treatment Phase of up to 1 year. On treatment discontinuation, patients will be reviewed in an End of Treatment Visit, and then 12 weekly from the end of treatment until disease progression. Following disease progression, patients will revert to routine local follow up processes.
Trial Status:
Recruiting
Population:
33-36 patients aged ≥18 years with incurable histologically confirmed, immunotherapy naïve urinary tract squamous cell carcinoma.

This trial is funded by Cancer Research UK (award reference no. CRCPJT\100018)
Senior Trial Manager:
Sarah-Jane Bibby
Trial Coordinator:
Mary Danh
Trial Assistant:
Armando Lobete-Ejeleri
Data Manager:
Zoe Konn
Clinical Data Coordinator:
Uroosha Ali Babul Hussain
Clinical Trial Monitor:
Oli Dewane
Contact Information for trial queries:
Email: aurora@soton.ac.uk
Phone: 023 8120 5154
SAE Reporting:
Email: ctu@soton.ac.uk
Investigator Site Files
00 AURORA Investigator Site File Index v1 18-MAR-2022
2.1 AURORA Master Patient List v1 15-FEB-2022
2.2 AURORA Screening Log v1 17-FEB-2022
3.1 AURORA Site Delegation Log v1 15-FEB-2022
3.3 AURORA Site Training Log Template v1 15-FEB-2022
4.1 AURORA Protocol v6 09-NOV-2023
4.3 AURORA Patient Information Sheet v5 09-NOV-2023
4.3 AURORA Pregnancy Patient Information Sheet v3 22-JUL-2022
4.4 AURORA Informed Consent Form v3 22-JUL-2022
4.4 AURORA - Pregnant Partner ICF v2 28-APR-2022
4.5 AURORA GP Letter v1 26-NOV-2021
4.7 - AURORA EORTC QLQ-C30 English
5.1 AURORA Instructions for Adverse and Serious Adverse Event Reporting v1 15-FEB-2022
5.2 AURORA SAE Report Form v3 21-MAR-2023
6.1 AURORA RO5541267 Atezolizumab - IB Version 19
6.3 AURORA Atezolizumab 840mg Accountability Log v1 15-FEB-2022
6.4 AURORA Atezolizumab Dispensing Log v1 15-FEB-2022
8.3 AURORA Sample Shipment Form for AURORA Archival Tumour Sample v4 08-JAN-2024
8.3 AURORA Sample Shipment Form for AURORA Streck Blood Samples v4 08-JAN-2024
8.3 AURORA Sample Shipment Form for AURORA Flow Whole Blood EDTA v4 08-JAN-2024
8.5 AURORA Lab Manual v4 08-JAN-2024
10.1 AURORA Letter_of_HRA_Approval_25.05.2022 20220525T151345.343 GMT_HRA-IRAS-WORK R-1583
12.1 AURORA CTA Acceptance of Amended Request 20220524T142640.385 GMT_HRA-IRAS-WORK P-1004493
13.1 AURORA_eCRF Completion Guidelines v4 26-JAN-2024
14.2 AURORA Site Visit Log v1 15-FEB-2022
Pharmacy Files
0. AURORA Pharmacy File Index v1 18-MAR-2022
3.1 AURORA Site Delegation Log v1 15-FEB-2022
3.3 AURORA Site Training Log Template v1 15-FEB-2022
4.1 AURORA Protocol v6 09-NOV-2023
4.2 AURORA Pharmacy Manual v4 03-MAY-2023
5.1 AURORA RO5541267 Atezolizumab - IB Version 19
5.2 AURORA Atezolizumab 840mg Accountability Log v1 15-FEB-2022
5.3 AURORA Atezolizumab Dispensing Log v1 15-FEB-2022
5.4 AURORA Drug Delivery Request Form v2 21-Oct-2022
5.5 AURORA Atezolizumab CoCTS v1 09-Mar-2022
5.12 AURORA Unblinding Reports Note to File v1 25-MAR-2022
9.1 CTA Acceptance of Amended Request 20220524T142640.385 GMT_HRA-IRAS-WORK P-1004493
Simon Crabb, Robin Wickens, Sarah Jane-Bibby, et al. Evaluating atezolizumab in patients with urinary tract squamous cell carcinoma (AURORA): study protocol for a single arm, open-label, multicentre, phase II clinical trial. BMC Cancer, 2023. doi.10.1186/s12885-023-11397-x
Press releases, video clips and other external websites:
(University of Southampton cannot accept responsibility for external websites)
Watch a video on the AURORA trial: