Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Trial Overview
Trial Team
Essential Trial Documentation
Publications and Presentations
Other Media
Title
Two single arm, multicentre unblinded first-in-human trials, including 2 phases and a qualitative sub study investigating a novel ureteric stent in Kidney stone patients and Oncology patients to determine the reduction of encrustation, biofilm deposition and complications compared to a conventional JJ stent (CASSETTE)
Description
A key problem with ureteric stents is that they can become narrowed or blocked by a build-up of calcium (like scaling in your kettle) or by bacteria (like the gunge in the plughole of your sink or bath). When this happens, patients may develop a urine infection, which needs to be treated with antibiotics. If a patient has many infections, the bacteria can become resistant to antibiotics, so that it becomes more difficult to treat the infections. If the stent is completely blocked, patients need to return to hospital for another operation to replace the stent.
Researchers in the Department of Mechanical Engineering at the University of Southampton have designed a new stent, which should be less prone to blockages. It has holes in the side with unique designs to improve the flow of urine and to prevent the build-up of calcium and bacteria. So,
- these stents should last longer before they need to be replaced.
- patients should need to come to hospital less often to have the stent changed, which should make their lives easier.
In this study, we want to test these stents in humans for the first time. We need to prove whether they are safe and acceptable to patients, and whether they work as well as we predict.
Objectives
Primary Objectives:
- To determine rates of stent failure
Secondary Objectives:
- To assess extent of encrustation and biofilm (E&B)
- To determine whether the novel stent leads to better clinical outcomes
- To assess impact of the novel ureteric stent on quality of life
- To understand patient experience in having the novel ureteric stent inserted and reason for participation in the trial
Exploratory:
- To assess extent of encrustation and biofilm (E&B)
Trial Design
Two single arm, multicentre unblinded first-in-human trials, including 2 phases and a qualitative sub study
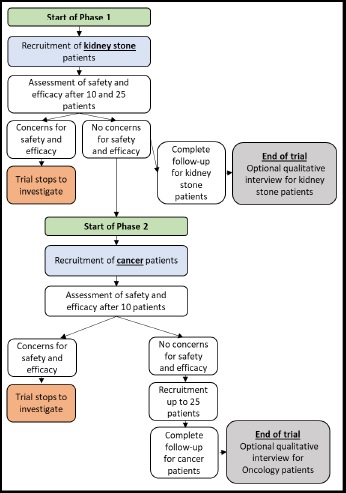
Trial Status:
In set-up
Population:
Patients (male & female) aged 18 years or older with kidney stones or abdominal cancers, with previous experience of ureteric stents and clinical indication for ureteric stents.
.jpg)
This trial is primarily funded by the National Institute for Health and Care Research (NIHR) Invention for Innovation (I4I) Product Development Award (PDA) programme. NIHR202935
Senior Trial Manager:
Zina Eminton
Trial Manager:
Sinead Helyar
Supporting Trial Manager:
Sophie Varkonyi-Clifford
Trial Monitor:
Parys Hatchard
Data Manager:
Emma Tilt
Contact Information for trial queries:
Email: cassette@soton.ac.uk
Phone: 023 8120 5154
SAE Reporting:
Email: ctu@soton.ac.uk
Patient facing documents
CASSETTE - Patient PIS - Main (Kidney Patients) v4 - 28-Apr-2025
CASSETTE - Patient PIS - Main (Oncology patients) v4 - 28-Apr-2025
CASSETTE - Patient PIS (Interview) v3 28-Apr-2025
CASSETTE - Clinician PIS (Interview) v3 - 28-Apr-2025
CASSETTE - Pregnancy Patient Information Sheet v2 - 28-Apr-2025
CASSETTE - Pregnancy informed consent form V3 28-Apr-2025
CASSETTE - Patient invitation letter v1 -22-Jan-2024
CASSETTE - Patient ICF (Sample collection and questionnaires) v1 13-Mar-2024
CASSETTE - Patient contact details consent_v1 13-Mar-2024
Sample management
CASSETTE - Device Shipment Form v1 28-Mar-2024
CASSETTE - Device Accountability v2 09-Apr-2025
CASSETTE - Laboratory Manual v2 15-Apr-2025
CASSETTE - Sample Shipment Form v1 22-May-2024
Miscellaneous
CASSETTE - GP Information Sheet v1 22-Jan-2024
Coming soon.
Press releases, video clips and other external websites:
(University of Southampton cannot accept responsibility for external websites)
