Breaking Barriers in Skin Sensing Assessment
This research evaluates the efficacy of using a novel electrical measurement technique for real-time assessment of skin barrier function for at risk patient demographics principally incontinence and diabetes.
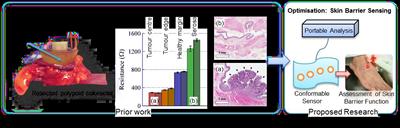
The research builds on, and translates, our prior work developing a galvanostatic measurement technique for tissue health assessment in colorectal cancer. This clearly demonstrated the potential of the technique for fast (<1s), robust and sensitive measures of tissue health using human tissue from colorectal cancer cases. The system comprises a measurement probe with 4 electrodes which contacts with the target tissue, linked to a compact data acquisition device and laptop for analysis. Although the clinical application differs, the technique measures electrochemical properties which are fundamentally linked to the cellular composition of the target tissue and thus directly affected by the onset of disease. Our working hypothesis is that we can therefore translate this technology to identify at-risk patient demographics facilitating the stratification of care both in-institution and community informed by a rapid, but sensitive measurement method.
The project comprises two phases; firstly a technical development phase will adapt our existing probe (which contacts the target tissue) into an appropriate form for testing on curved skin surfaces, secondly this revised system will be used in a small pilot study with diabetes patients at Leeds Community Healthcare NHS Trust:
Phase 1: Our existing probe is time-consuming to fabricate and rigid (since internal tissues conform to its surface). We will exploit recent advances in flexible electrode manufacture at University of Leeds (through our EPSRC Robotics Fabrication centre) to fabricate compliant probes with copper-electrodes for direct application to skin tissue, thus enabling future integration with bandages or dressings. These electrodes will be characterised and calibrated using ex vivo porcine skin tissue.
Phase 2: We will evaluate the revised system developed in Phase 1 in a small study of patients with diabetic foot. A cohort of 10 patients with moderate symptoms will be recruited and the measurement system will be used to measure locations spatially distributed across each patient’s foot. A clinician will supervise and guide the study. In each subject the clinician will identify 10 healthy and diseased locations, 5 repeat measures will be taken from each location. The resultant data will be analysed statistical techniques to characterise the ability of the technique to discriminate changes in tissue health within and between subjects.